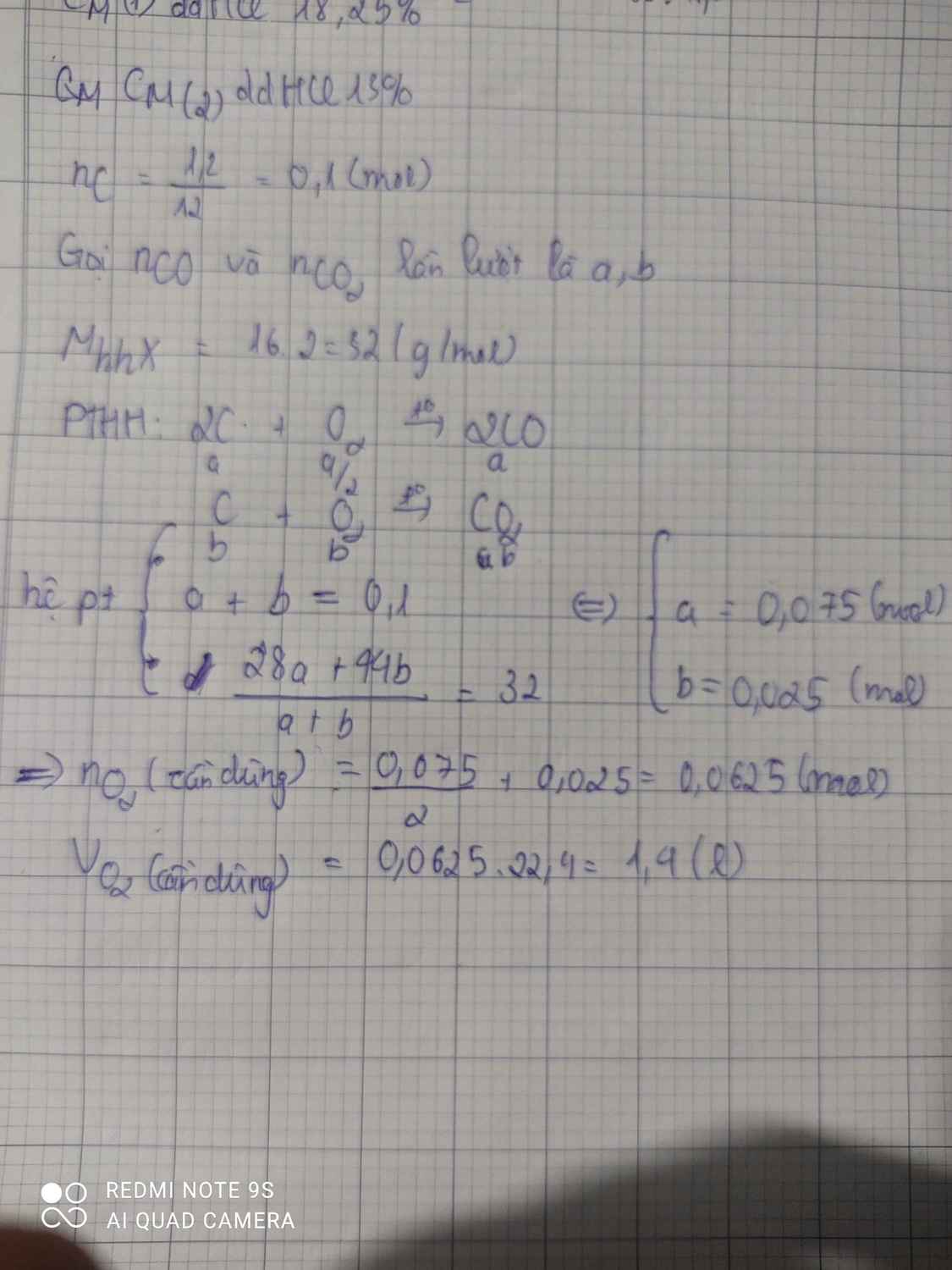\(n_C=\dfrac{1,2}{12}=0,1\left(mol\right)\)
PTHH: C + O2 --to--> CO2
a-->a--------->a
2C + O2 --to--> 2CO
b--->0,5b------>b
=> a + b = 0,1
Có: \(\overline{M}_X=\dfrac{44a+28b}{a+b}=16.2=32\)
=> a = 0,025; b = 0,075
\(n_{O_2}=a+0,5b=0,0625\left(mol\right)\)
=> \(V_{O_2}=0,0625.22,4=1,4\left(l\right)\)