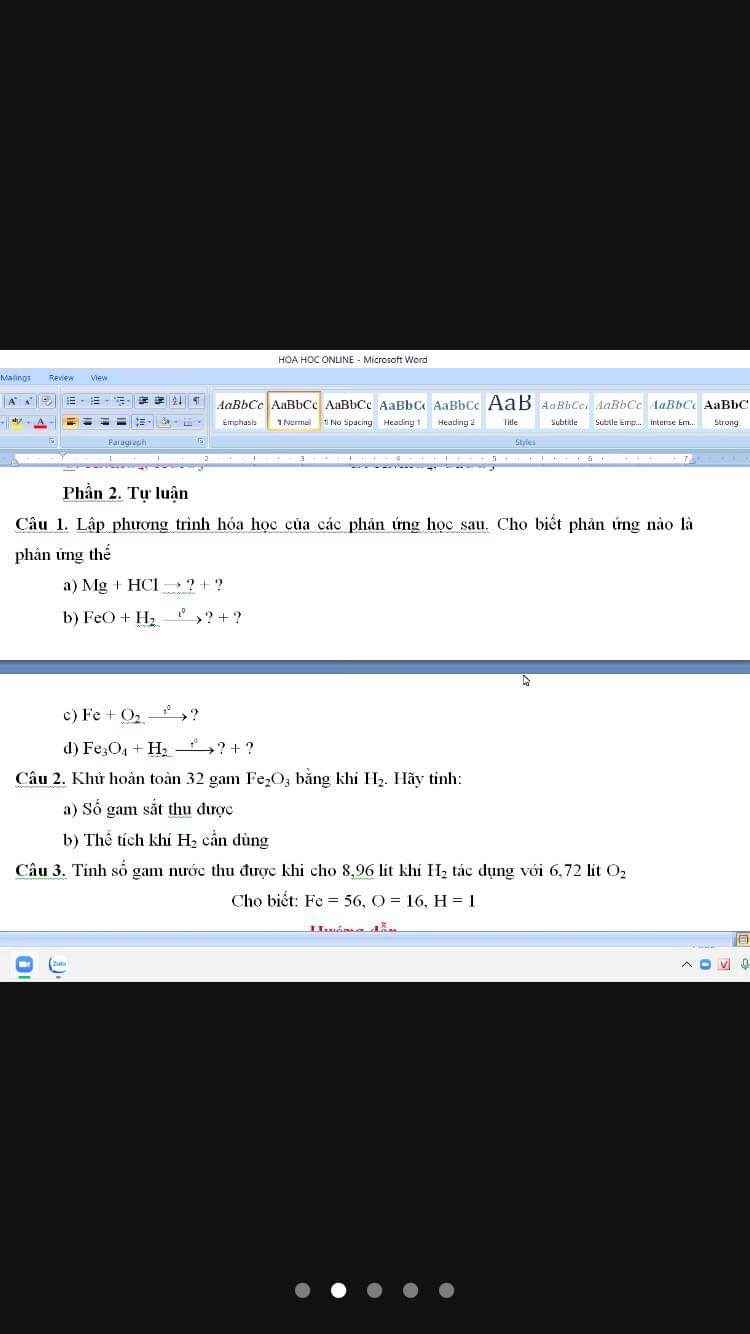
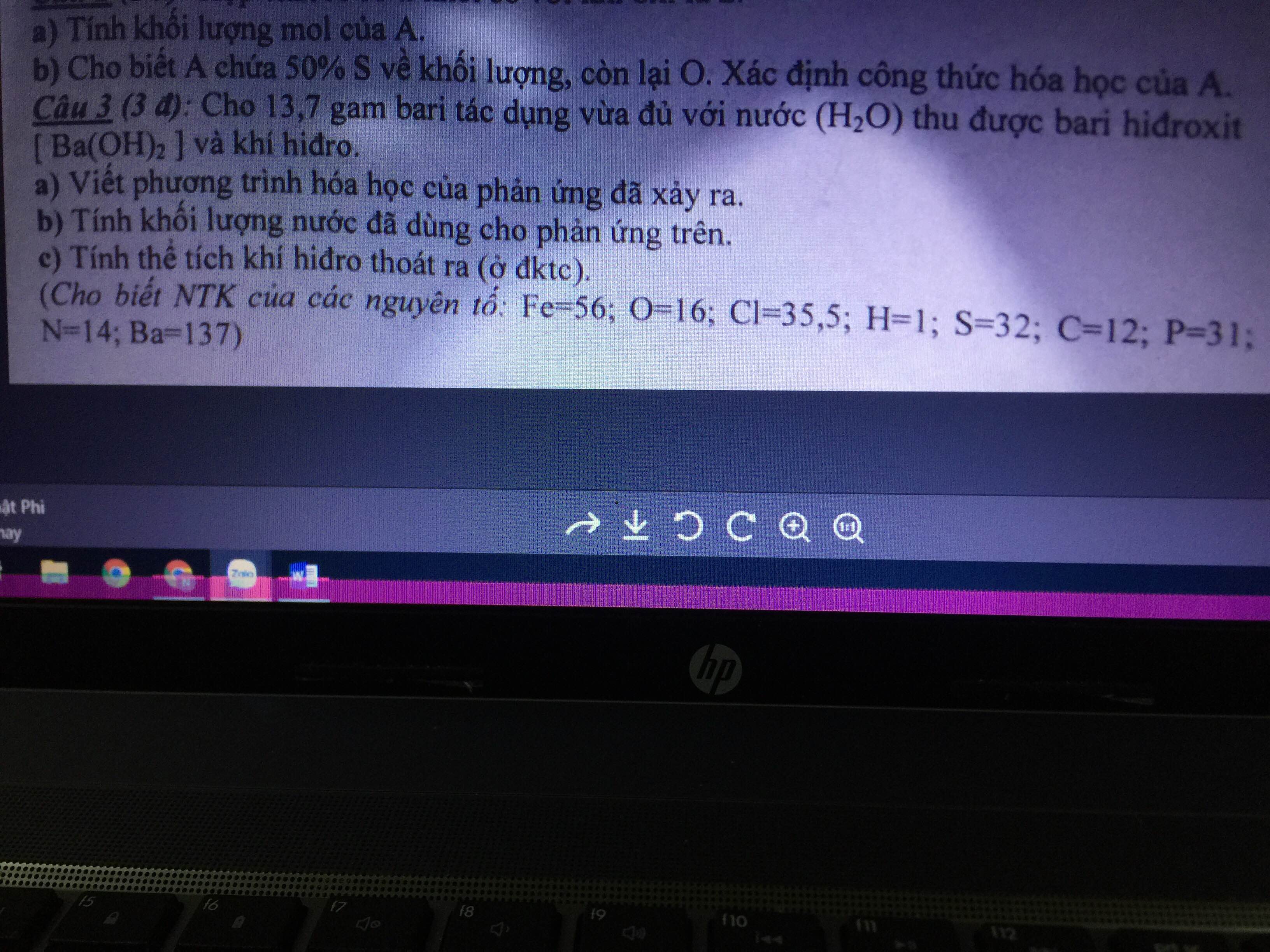
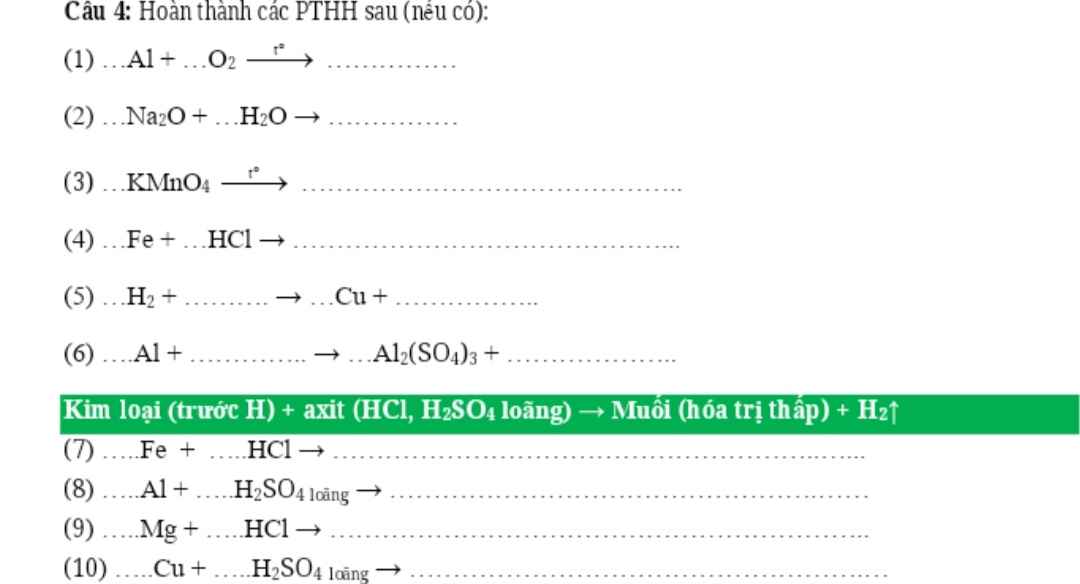
Câu 1:
a, \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
b, \(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
Theo PT: \(n_{H_2}=\dfrac{3}{2}n_{Al}=0,3\left(mol\right)\Rightarrow V_{H_2}=0,3.22,4=6,72\left(l\right)\)
c, \(n_{HCl}=3n_{Al}=0,6\left(mol\right)\)
\(\Rightarrow C\%_{HCl}=\dfrac{0,6.36,5}{200}.100\%=10,95\%\)
d, \(n_{AlCl_3}=n_{Al}=0,2\left(mol\right)\Rightarrow m_{AlCl_3}=0,2.133,5=26,7\left(g\right)\)