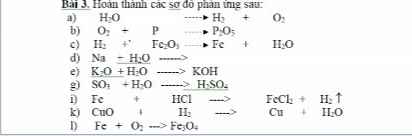
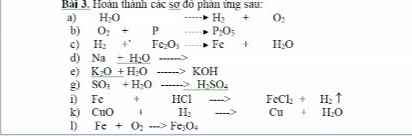
Câu 1 :
$M + O_2 \xrightarrow{t^o} MO_2$
Theo PTHH : $n_M = n_{MO_2} \Rightarrow \dfrac{2}{M} = \dfrac{2,54}{M + 32}$
$\Rightarrow M = 118,5 \to$ Sai đề
Câu 2 :
$2Ba + O_2 \xrightarrow{t^o} 2BaO$
$n_{O_2} = \dfrac{1}{2}n_{BaO} = 0,5.\dfrac{36,72}{153} = 0,12(mol)$
$4Fe(NO_3)_3 \xrightarrow{t^o} 2Fe_2O_3 + 12NO_2 + 3O_2$
$n_{Fe(NO_3)_3} = \dfrac{4}{3}n_{O_2} = 0,16(mol)$
$m_{Fe(NO_3)_3} = 0,16.242 = 38,72(gam)$
Câu 3 :
$2CO + O_2 \xrightarrow{t^o} 2CO_2$
$2H_2 + O_2 \xrightarrow{t^o} 2H_2O$
$V_{CO} = V_{CO_2} = 3,36(lít)$
$V_{CO} + V_{H_2} = 2V_{O_2} = 8,96 \Rightarrow V_{H_2} = 5,6(lít)$
$\%V_{CO} = \dfrac{3,36}{8,96}.100\% = 37,5\%$
$\%V_{H_2} = 100\% -37,5\% = 62,5\%$