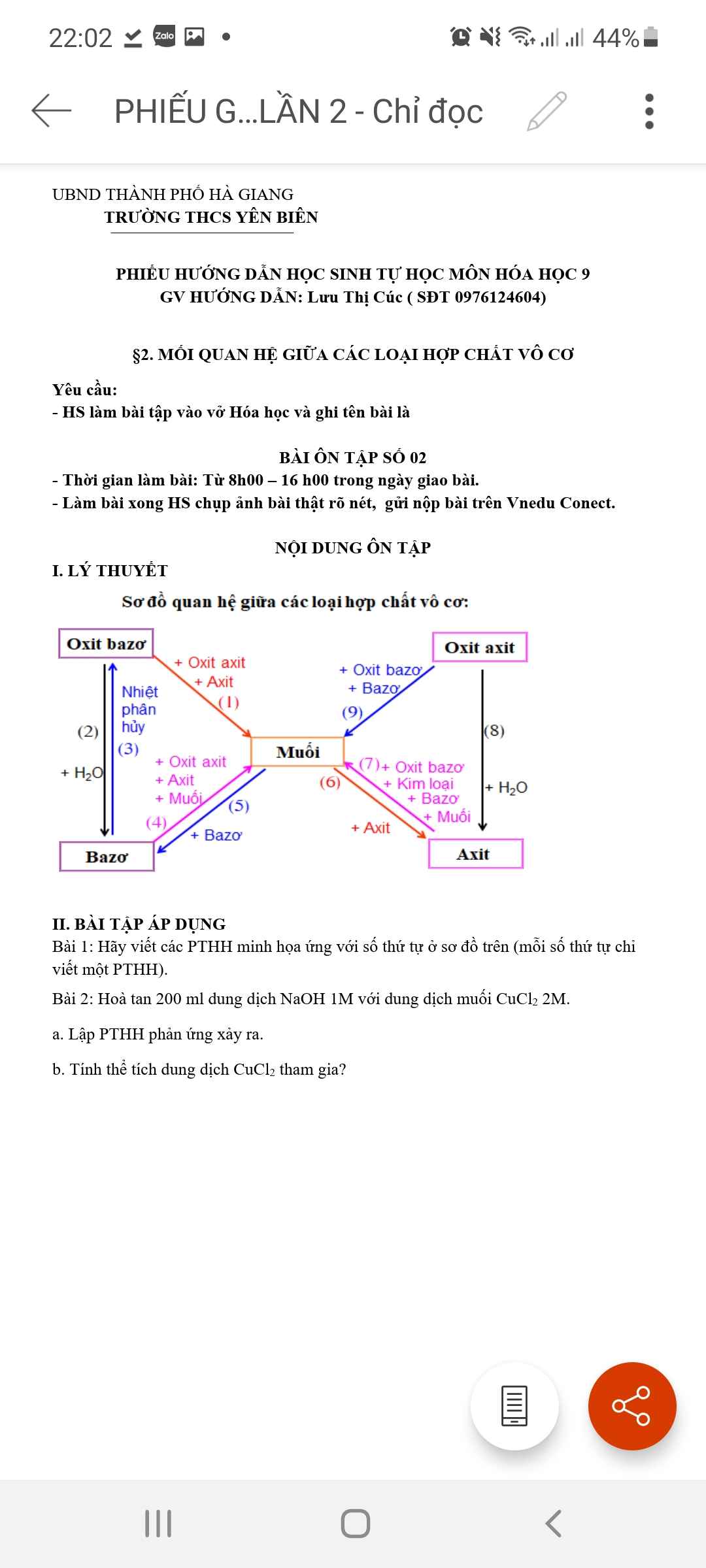
1) \(n_{Na_2SO_4}=0,2.1=0,2\left(mol\right)\)
PTHH: Na2SO4 + BaCl2 → 2NaCl + BaSO4 ↓
Mol: 0,2 0,2 0,2 0,2
\(m_{BaSO_4}=0,2.233=46,6\left(g\right)\)
2) \(m_{NaCl}=0,2.58,5=11,7\left(g\right)\)
3) \(C_{M_{ddBaCl_2}}=\dfrac{0,2}{0,3}=\dfrac{2}{3}\approx0,67M\)
4) Vdd sau pứ = 0,2 + 0,3 = 0,5 (l)
\(C_{M_{ddNaCl}}=\dfrac{0,2}{0,5}=0,4M\)
5) mdd sau pứ = 500.1,101 = 550,5 (g) (0,5l = 500ml)
\(C\%_{ddNaCl}=\dfrac{11,7.100\%}{550,5}=2,12534\%\)