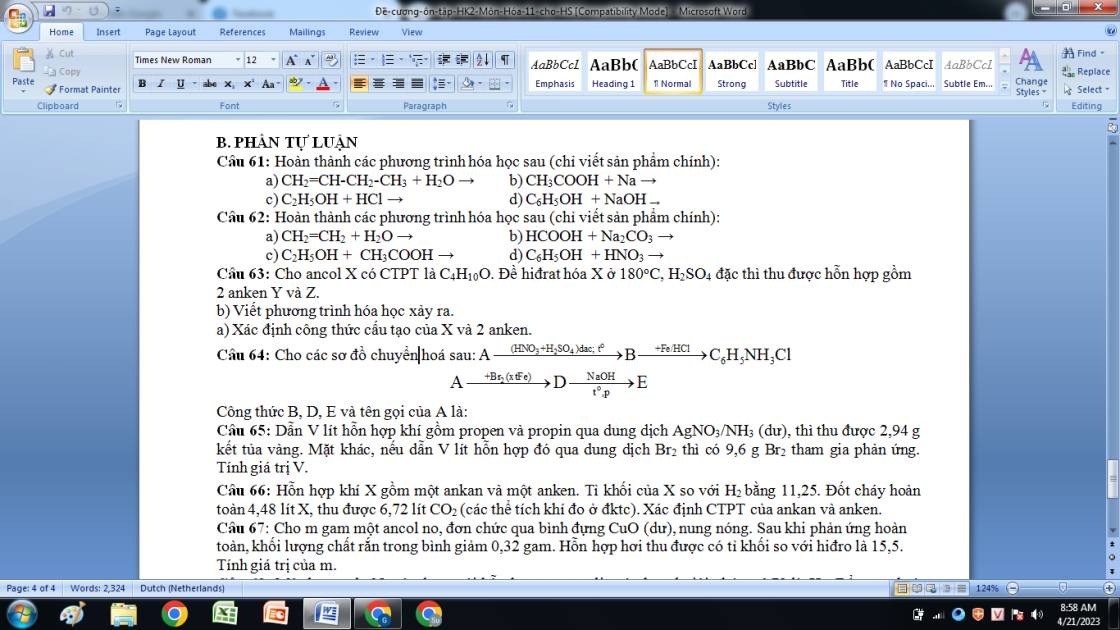
Bài 6:
\(n_{C_2Ag_2}=\dfrac{24}{240}=0,1\left(mol\right)\)
=> nC2H2 = 0,1 (mol)
Khí thoát ra khỏi dd Br2 là C2H6
\(n_{C_2H_6}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
=> \(n_{C_2H_4}=\dfrac{6,72}{22,4}-0,1-0,1=0,1\left(mol\right)\)
\(\left\{{}\begin{matrix}m_{C_2H_2}=0,1.26=2,6\left(g\right)\\m_{C_2H_4}=0,1.28=2,8\left(g\right)\\m_{C_2H_6}=0,1.30=3\left(g\right)\end{matrix}\right.\)
Bài 7:
\(n_{C_3H_3Ag}=\dfrac{22,05}{147}=0,15\left(mol\right)\)
=> nC3H4 = 0,15 (mol)
Khí thoát ra khỏi binh đựng Br2 là C2H6
\(n_{C_2H_6}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
=> \(n_{C_2H_4}=\dfrac{8,96}{22,4}-0,15-0,1=0,15\left(mol\right)\)
\(\left\{{}\begin{matrix}m_{C_2H_4}=0,15.28=4,2\left(g\right)\\m_{C_2H_6}=0,1.30=3\left(g\right)\\m_{C_3H_4}=0,15.40=6\left(g\right)\end{matrix}\right.\)
Bài 7:
nC2H6=2,2422,4=0,1(mol)nC2H6=2,2422,4=0,1(mol)
=>