\(a,PTHH:4Al+3O_2\underrightarrow{t^o}2Al_2O_3\\ b,n_{Al}=\dfrac{2,7}{27}=0,1\left(mol\right)\\ Theo.pt:n_{Al_2O_3}=\dfrac{1}{2}n_{Al}=\dfrac{1}{2}.0,1=0,05\left(mol\right)\\ m_{Al_2O_3}=0,05.102=5,1\left(g\right)\\ c,THeo.pt:n_{O_2}=\dfrac{3}{4}n_{Al}=\dfrac{3}{4}.0,1=0,075\left(mol\right)\\ V_{kk}=0,075.22,4.5=8,4\left(l\right)\)
Đúng 3
Bình luận (0)







 A-Ai cứu mình với...bài nhiều quá...mình không làm nổi...Mấy bạn giúp mình với...
A-Ai cứu mình với...bài nhiều quá...mình không làm nổi...Mấy bạn giúp mình với...







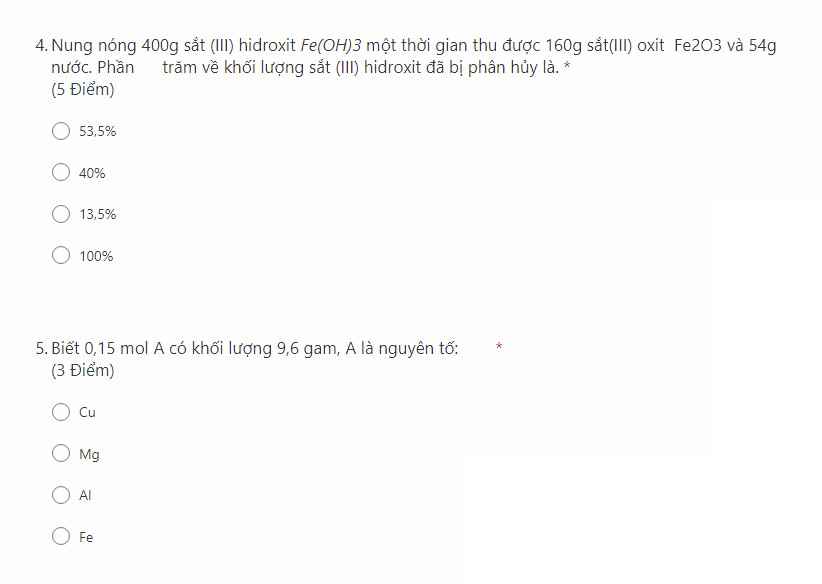
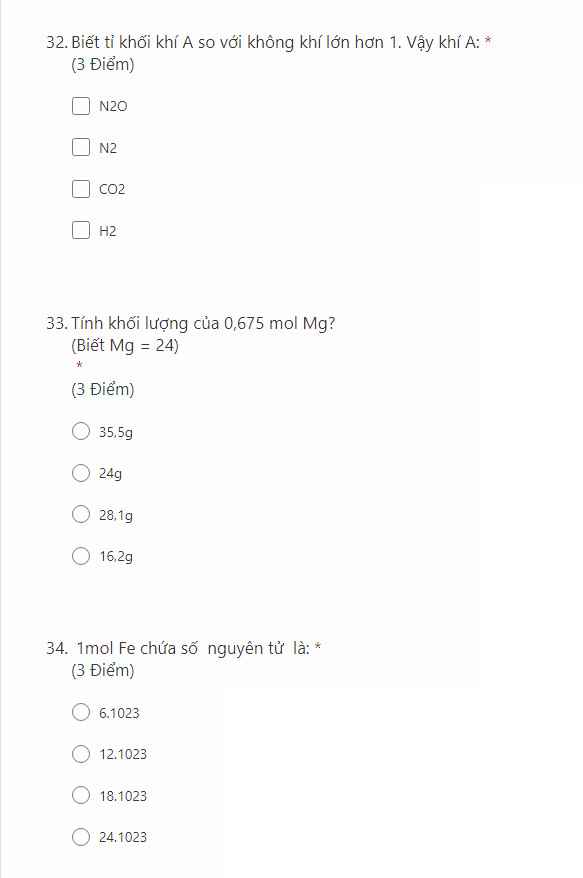

 ai giúp mình với ạ
ai giúp mình với ạ 