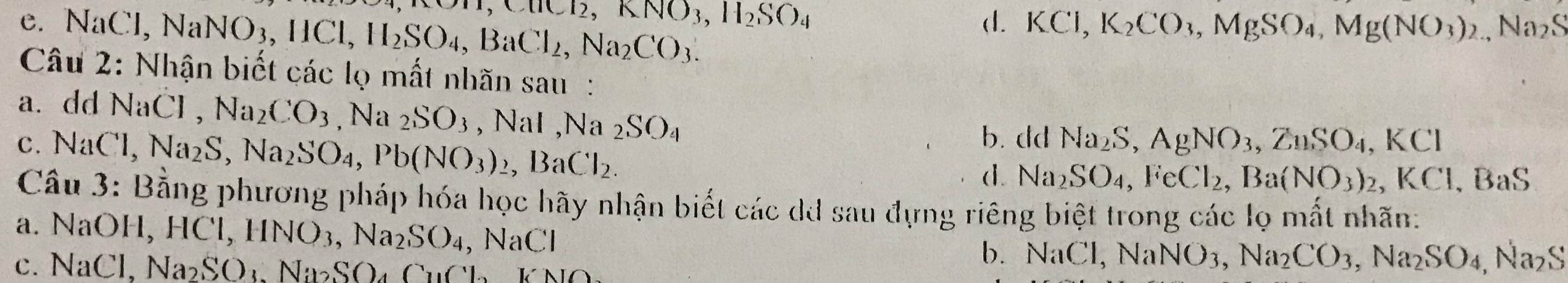Câu 29:
(1) \(Cl_2+2NaOH\rightarrow NaCl+NaClO+H_2O\)
(2) \(2Fe+3Cl_2\underrightarrow{t^o}2FeCl_3\)
(3) \(H_2+Cl_2\underrightarrow{t^o}2HCl\)
(4) \(HCl+NaOH\rightarrow NaCl+H_2O\)
(5) \(NaCl+AgNO_3\rightarrow NaNO_3+AgCl_{\downarrow}\)
(6) \(Fe+2HCl\rightarrow FeCl_2+H_2\)
Câu 30:
a, PT: \(Zn+S\underrightarrow{t^o}ZnS\)
\(Fe+S\underrightarrow{t^o}FeS\)
b, Giả sử: \(\left\{{}\begin{matrix}n_{Zn}=x\left(mol\right)\\n_{Fe}=y\left(mol\right)\end{matrix}\right.\)
⇒ 65x + 56y = 26,55 (1)
Ta có: \(n_S=\dfrac{14,4}{32}=0,45\left(mol\right)\)
Theo PT: \(n_S=n_{Zn}+n_{Fe}=x+y\left(mol\right)\)
⇒ x + y = 0,45 (2)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}x=0,15\left(mol\right)\\y=0,3\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{0,15.65}{26,55}.100\%\approx36,7\%\\\%m_{Fe}\approx63,3\%\end{matrix}\right.\)
Bạn tham khảo nhé!