Quy hết kim loại R (II) và Fe về M (II):
\(n_{HCl}=0,2.2=0,4\left(mol\right)\)
PTHH: M + 2HCl ---> MCl2 + H2↑
0,2<-0,4------->0,2---->0,2
=> VH2 = 0,2.22,4 = 4,48 (l)
\(b,M_M=\dfrac{8}{0,2}=40\left(\dfrac{g}{mol}\right)\\ \rightarrow M_R=2.40-56=24\left(\dfrac{g}{mol}\right)\)
=> R là Mg
c, Gọi \(\left\{{}\begin{matrix}n_{Fe}=a\left(mol\right)\\n_{Mg}=b\left(mol\right)\end{matrix}\right.\left(a,b>0\right)\)
PTHH:
Fe + 2HCl ---> FeCl2 + H2
a---->2a--------->a
Mg + 2HCl ---> MgCl2 + H2
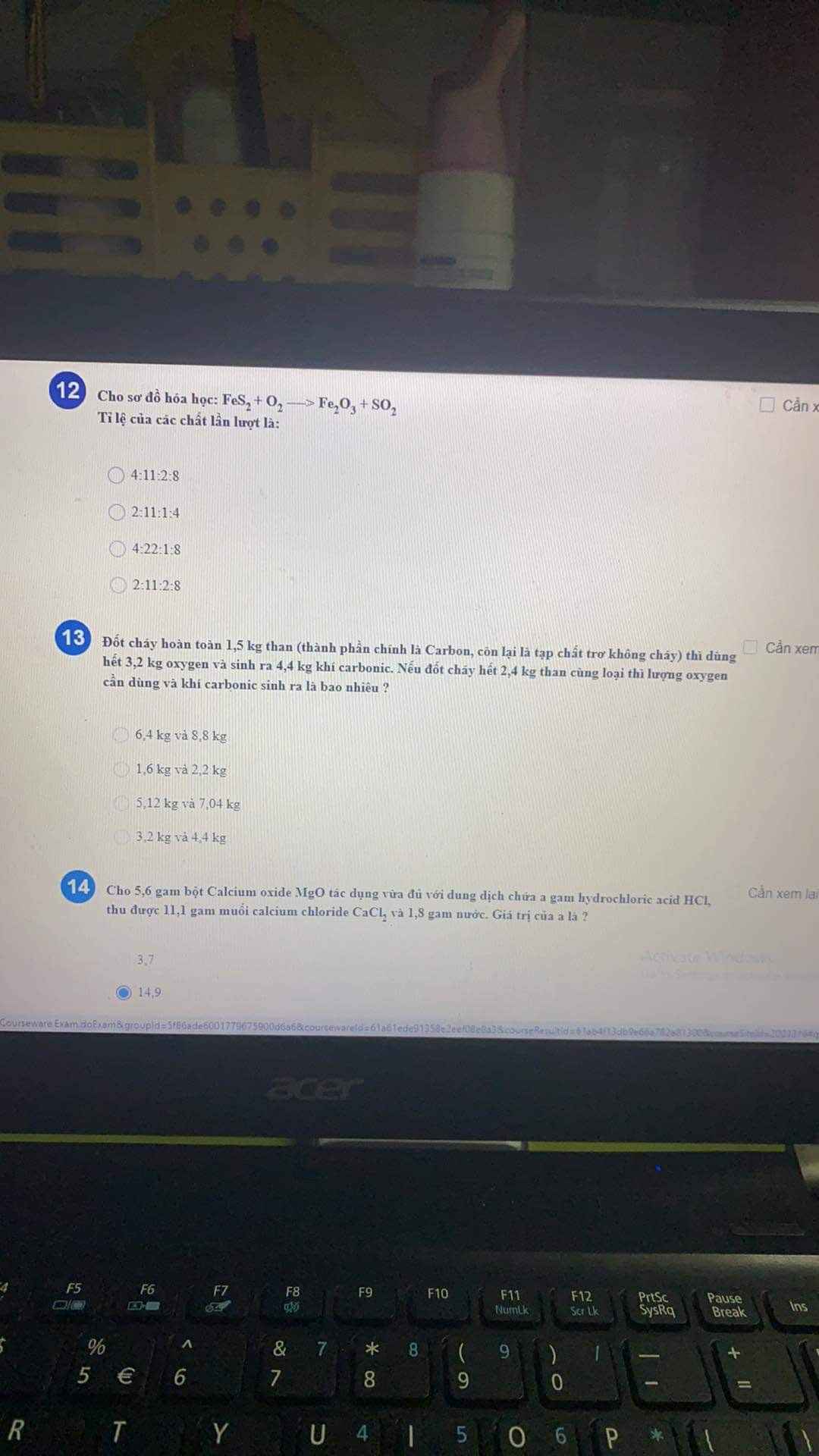
b---->2b-------->b
=> hệ pt \(\left\{{}\begin{matrix}56a+24b=8\\2a+2b=0,4\end{matrix}\right.\Leftrightarrow a=b=0,1\left(mol\right)\left(TM\right)\)
=> \(\left\{{}\begin{matrix}m_{Fe}=0,1.56=5,6\left(g\right)\\m_{Mg}=0,1.24=2,4\left(g\right)\end{matrix}\right.\)
d, \(\left\{{}\begin{matrix}C_{M\left(FeCl_2\right)}=\dfrac{0,1}{0,2}=0,5M\\C_{M\left(MgCl_2\right)}=\dfrac{0,1}{0,2}=0,5M\end{matrix}\right.\)
Gọi kim loại cần tìm là R (II)
Gọi \(\left\{{}\begin{matrix}n_{Fe}=a\left(mol\right)\\n_R=b\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow56a+M_R.b=8\left(g\right)\)
\(n_{HCl}=0,2.2=0,4\left(mol\right)\)
PTHH:
Fe + 2HCl --->FeCl2 + H2
a---->2a
R + 2HCl ---> RCl2 + H2
b---->2b
Theo pthh: \(n_{hhkl}=n_{H_2}=\dfrac{1}{2}n_{HCl}=\dfrac{1}{2}.0,4=0,2\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
b, Lập được hệ pt: \(\left\{{}\begin{matrix}56a+M_R.b=8\\2a+2b=0,4\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,1\left(mol\right)\\b=0,1\left(mol\right)\\M_R=24\left(\dfrac{g}{mol}\right)\end{matrix}\right.\)
=> R là Mg
c, \(\left\{{}\begin{matrix}n_{Fe}=0,1\left(mol\right)\\n_{Mg}=0,1\left(mol\right)\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}m_{Fe}=0,1.56=5,6\left(g\right)\\m_{Mg}=0,1.56=5,6\left(g\right)\end{matrix}\right.\)








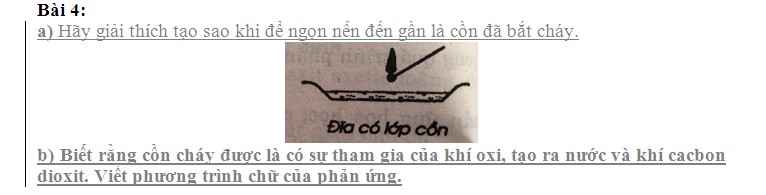 mấy anh chị giải chi tiết giùm em,em cần lời giải chi tiết ạ em xin cảm ơn
mấy anh chị giải chi tiết giùm em,em cần lời giải chi tiết ạ em xin cảm ơn


 giải chi tiết giúp mình c4 ạ. Mình cảm ơn!!!!!!!!
giải chi tiết giúp mình c4 ạ. Mình cảm ơn!!!!!!!!