\(n_P=0,25\left(mol\right);n_{O_2}=0,25\left(mol\right)\\ 4P+5O_2-^{t^o}\rightarrow2P_2O_5\\ LTL:\dfrac{0,25}{4}>\dfrac{0,25}{5}\Rightarrow Pdư\\ n_{P\left(dư\right)}=0,25-\dfrac{0,25.4}{5}=0,05\left(mol\right)\\n_{P_2O_5}=\dfrac{2}{5}n_{O_2}=0,1\left(mol\right)\\ m_{P_2O_5}=0,1.142=14,2\left(g\right) \)
Đề cương ôn tập cuối HKI
Đúng 1
Bình luận (0)
Các câu hỏi tương tự
 giúp mình bài 7, bài 8 với ạ, cảm ơn nhiều
giúp mình bài 7, bài 8 với ạ, cảm ơn nhiều
Giúp mình bài 4 với ạ :( 
Mọi người giúp mình giải 2 phần này vs ☺😟😟😟😟Mk cảm ơn trước ạ :))))

Hãy tính:
a) Số mol của: 19,6g H2SO4; 3×10^23 phân tử CO2; 1,12 lít khí O2(đvtc)
b) Tính thể tích của hỗn hợp khí(đvtc) gồm có: 5,6g N2 và 1,8×10^23 phân tử O2
Mọi người giúp em nhanh và ạ.
Chỉ giúp với ạ


Giúp mik với ạ!!!!!!!!!
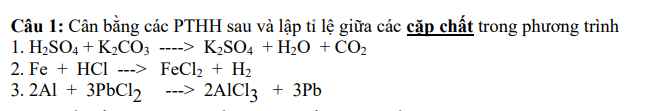
Giúp mik dc ko ạ
Cho 13g kém tác dụng hết vớ đ axit clohdric HCl 14,6% sau phản ứng thu được kém clorua và khí hidro
A) Tính thể tích khí hidro sinh ra ở dktc
B) tính khối lượng đ HCl đã dùng
C) Dẫn toàn bộ khí sinh ra qua đồng (II) oxit nung nóng. Tính khối lượng đồng tạo thành sau phản ứng.
Mọi người giải giúp em nhanh nhanh ạ. Mai em nộp bài mà đọc kiểu câu hỏi có tính chất bắc cầu kiểu này em không rõ lắm, nên mọi người làm chi tiết giùm em chút được không ạ❤❤
Đọc tiếp
Cho 13g kém tác dụng hết vớ đ axit clohdric HCl 14,6% sau phản ứng thu được kém clorua và khí hidro
A) Tính thể tích khí hidro sinh ra ở dktc
B) tính khối lượng đ HCl đã dùng
C) Dẫn toàn bộ khí sinh ra qua đồng (II) oxit nung nóng. Tính khối lượng đồng tạo thành sau phản ứng.
Mọi người giải giúp em nhanh nhanh ạ. Mai em nộp bài mà đọc kiểu câu hỏi có tính chất bắc cầu kiểu này em không rõ lắm, nên mọi người làm chi tiết giùm em chút được không ạ❤❤
ai có đề cương ôn thi học kì 1 hóa học lớp 8 không ạ???
cho em xin mượn với
một câu cũng đc













