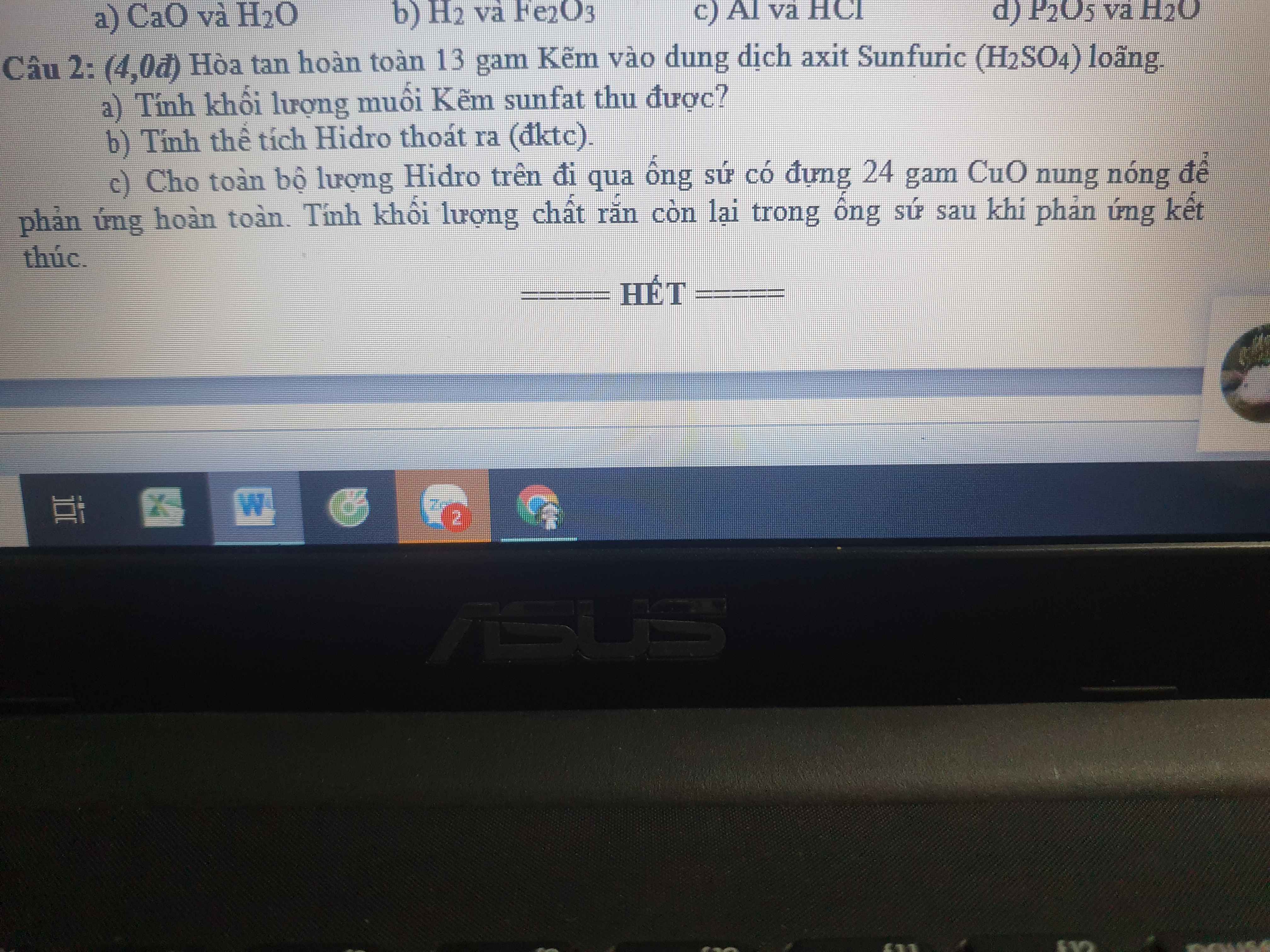
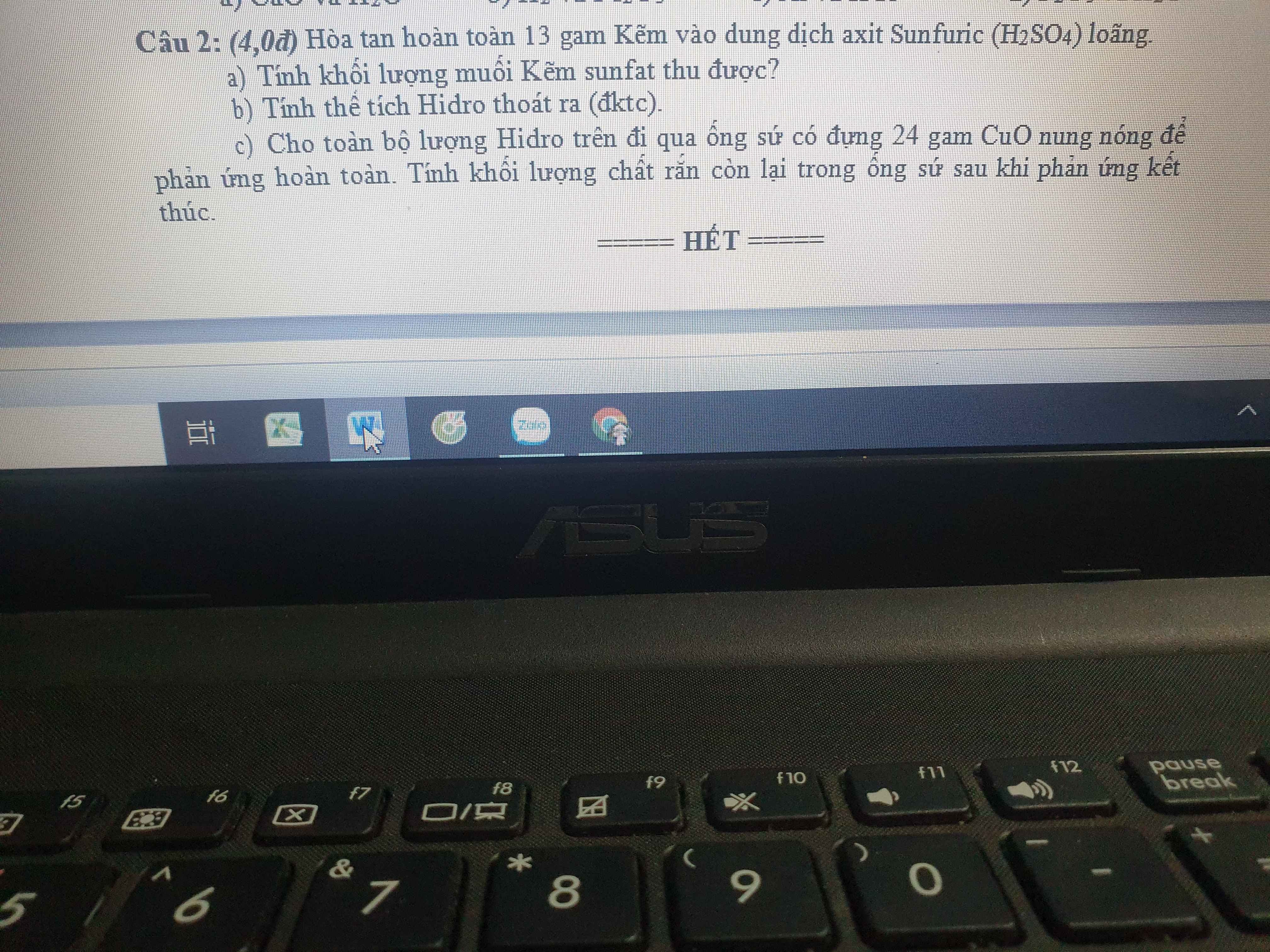
a) \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
PTHH: Zn + H2SO4 --> ZnSO4 + H2
0,2-------------->0,2----->0,2
=> \(m_{ZnSO_4}=0,2.161=32,2\left(g\right)\)
b) VH2 = 0,2.22,4 = 4,48 (l)
c) \(n_{CuO}=\dfrac{24}{80}=0,3\left(mol\right)\)
PTHH: CuO + H2 --to--> Cu + H2O
Xét tỉ lệ: \(\dfrac{0,3}{1}>\dfrac{0,2}{1}\) => H2 hết, CuO dư
PTHH: CuO + H2 --to--> Cu + H2O
0,2<---0,2------>0,2
=> mrắn sau pư = 24 - 0,2.80 + 0,2.64 = 20,8 (g)
\(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\\
pthh:Zn+H_2SO_4\rightarrow ZnSO_4+H_2\)
0,2 0,2 0,2
\(m_{ZnSO_4}=0,2.136=17,2\left(g\right)\\
V_{H_2}=0,2.22,4=4,48\left(l\right)\\
n_{CuO}=\dfrac{24}{80}=0,3\left(mol\right)\\
pthh:CuO+H_2\underrightarrow{t^o}Cu+H_2O\\
LTL:\dfrac{0,3}{1}>\dfrac{0,2}{1}\)
=> CuO dư
\(n_{CuO\left(p\text{ư}\right)}=n_{Cu}=n_{H_2}=0,2\left(mol\right)\\
m_{CuO\left(d\right)}=\left(0,3-0,2\right).80=8\left(g\right)\\
m_{Cr}=8+\left(0,2.64\right)=20,8\left(g\right)\)