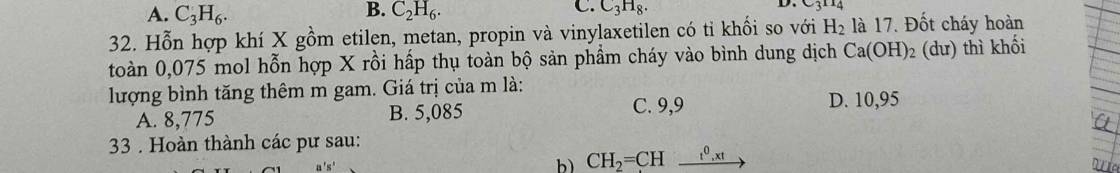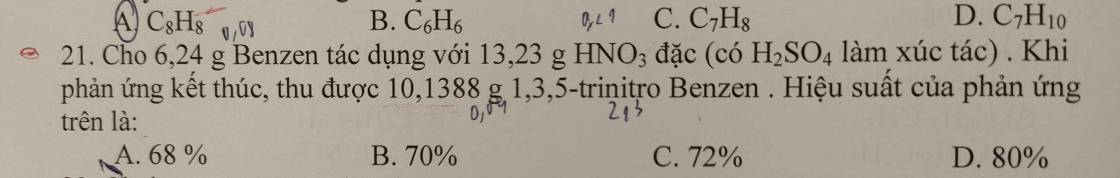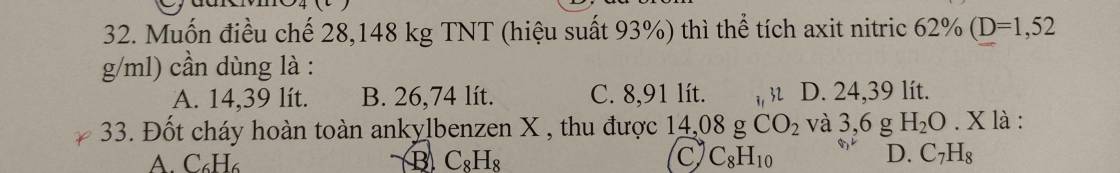\(M_X=17.2=34\left(g/mol\right)\)
Quy đổi hh \(X\left\{{}\begin{matrix}CH_4\\C_2H_4\\C_3H_4\\C_4H_4\end{matrix}\right.\rightarrow C_xH_4\left(1< x< 4\right)\)
\(m_X=34.0,075=2,55\left(g\right)\)
Mà \(n_H=4n_X=0,3\left(mol\right)\Rightarrow n_C=\dfrac{2,55-0,3}{12}=0,1875\left(mol\right)\)
BTNT: \(\left\{{}\begin{matrix}n_{CO_2}=n_C=0,1875\left(mol\right)\\n_{H_2O}=\dfrac{1}{2}n_H=0,15\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow m=m_{CO_2}+m_{H_2O}=0,1875.44+0,15.18=10,95\left(g\right)\)
Chọn D