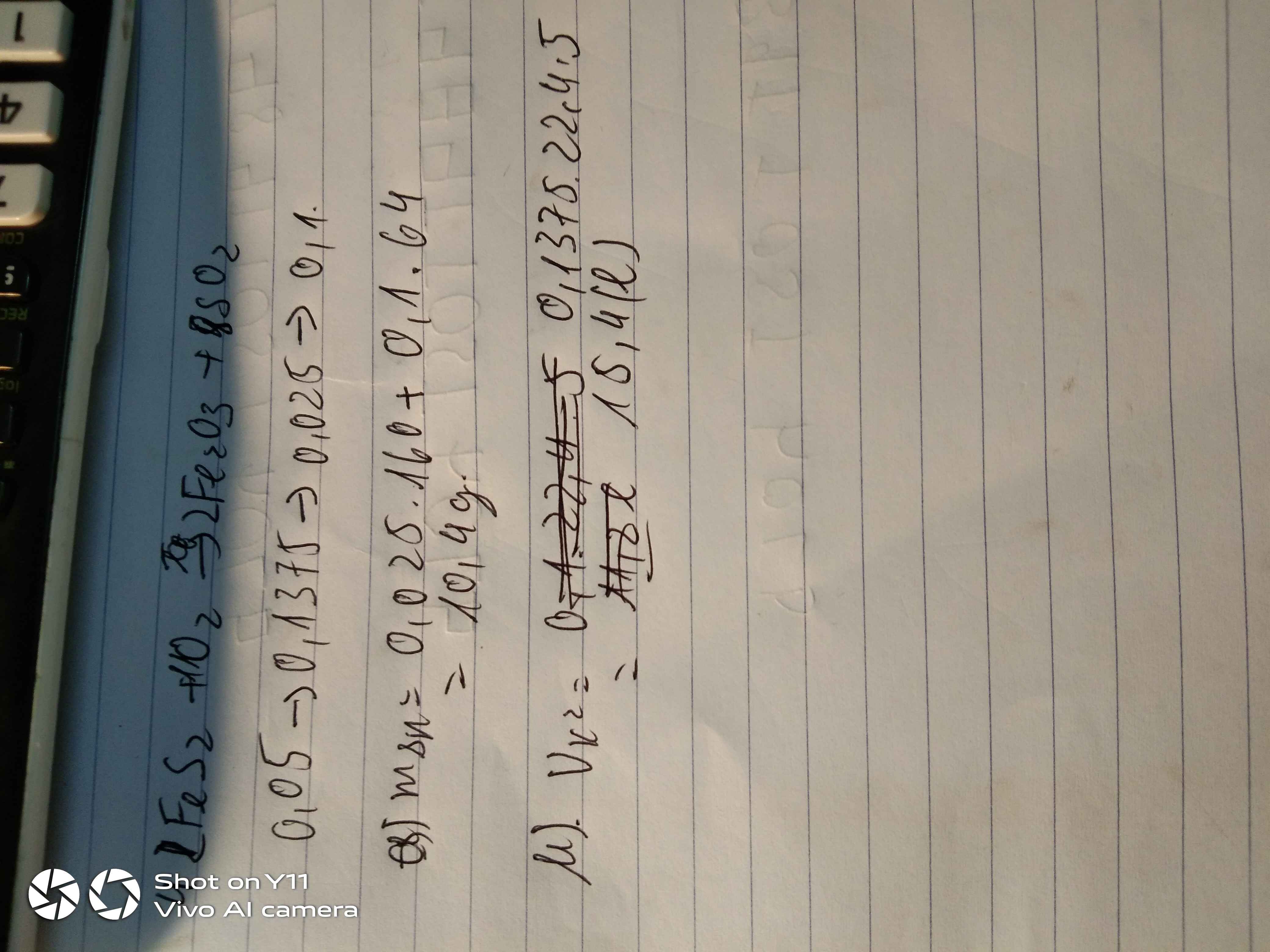\(a,n_{FeS_2}=\dfrac{m_{FeS_2}}{M_{FeS_2}}=\dfrac{6}{120}=0,05\left(mol\right)\\ 4FeS_2+11O_2\rightarrow\left(t^o,xt\right)2Fe_2O_3+8SO_2\uparrow\\ n_{Fe_2O_3}=\dfrac{2}{4}.n_{FeS_2}=\dfrac{2}{4}.0,05=0,025\left(mol\right)\\ \Rightarrow m_{Fe_2O_3}=160.0,025=4\left(g\right)\\ n_{SO_2}=\dfrac{8}{4}.n_{FeS_2}=\dfrac{8}{4}.0,05=0,1\left(mol\right)\\ \Rightarrow m_{SO_2}=0,1.64=6,4\left(g\right)\\ \Rightarrow m_{sp}=m_{Fe_2O_3}+m_{SO_2}=4+6,4=10,4\left(g\right)\\ b,n_{O_2}=\dfrac{11}{4}.n_{FeS_2}=\dfrac{11}{4}.0,05=0,1375\left(mol\right)\\ \Rightarrow V_{O_2\left(đktc\right)}=0,1375.22,4=3,08\left(l\right)\\ \Rightarrow V_{kk\left(đktc\right)}=3,08.5=15,4\left(l\right)\)
\(pthh:4FeS_2+11O_2\overset{t^o}{--->}2Fe_2O_3+8SO_2\uparrow\)
a. Ta có: \(n_{FeS_2}=\dfrac{6}{120}=0,05\left(mol\right)\)
Theo pt: \(n_{O_2}=\dfrac{11}{4}.n_{FeS_2}=\dfrac{11}{4}.0,05=0,1375\left(mol\right)\)
\(\Rightarrow m_{sản.phẩm.thu.được}=6+0,1375.32=10,4\left(g\right)\)
b. Ta có: \(V_{O_2}=0,1375.22,4=3,08\left(lít\right)\)
Mà: \(V_{O_2}=\dfrac{1}{5}V_{kk}\)
\(\Rightarrow V_{kk}=3,08.5=15,4\left(lít\right)\)
a: \(4FeS_2+11O_2\rightarrow2Fe_2O_3+8SO_2\uparrow\)
\(n_{FeS_2}=\dfrac{6}{129}\)(mol)
\(\Leftrightarrow n_{Fe_2O_3}=\dfrac{3}{129}\left(mol\right)\)
\(m_{Fe_2O_3}=\dfrac{3}{129}\cdot178=\dfrac{178}{43}\left(g\right)\)
b: \(n_{O_2}=\dfrac{2}{129}\left(mol\right)\)
\(V=n\cdot22.4=\dfrac{2}{129}\cdot22.4=\dfrac{224}{645}\left(lít\right)\)