a. \(PTHH:3H_2+Fe_2O_3\underrightarrow{t^o}2Fe+3H_2O\)
b. \(n_{Fe_2O_3}=\dfrac{m_{Fe_2O_3}}{M_{Fe_2O_3}}=\dfrac{4}{160}=0,025\left(mol\right)\)
\(PTHH:3H_2+Fe_2O_3\underrightarrow{t^o}2Fe+3H_2O\)
Mol : 3 : 1 : 2 : 3
Mol : 0,075 ← 0,025 → 0,05 → 0,075
\(\Rightarrow n_{H_2}=0,075\left(mol\right)\)
\(\Rightarrow V_{H_2}=n_{H_2}.22,4=0,075.22,4=1,68\left(l\right)\)
c. Từ câu b. \(\Rightarrow n_{Fe}=0,05\left(mol\right)\)
\(\Rightarrow m_{Fe}=n_{Fe}.M_{Fe}=0,05.56=2,8\left(g\right)\)










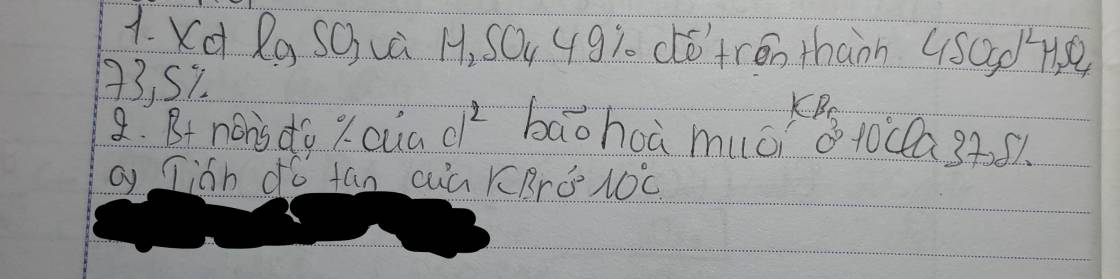 giải giúp mik vs ạ mik đang cần gấp!!!
giải giúp mik vs ạ mik đang cần gấp!!!


 các bn bỏ bài 16 , 17 , 21 nha Còn giúp mik vs nha mik đg cần gấp
các bn bỏ bài 16 , 17 , 21 nha Còn giúp mik vs nha mik đg cần gấp



