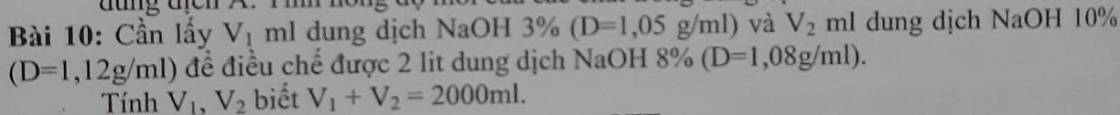Ta có: \(m_{ddNaOH8\%}=2000.1,08=2160\left(g\right)\)
\(n_{NaOH8\%}=\dfrac{2160.8\%}{40}=4,32\left(mol\right)\)
\(m_{ddNaOH3\%}=1,05V_1\left(g\right)\) \(\Rightarrow n_{NaOH3\%}=\dfrac{1,05V_1.3\%}{40}=7,875.10^{-4}V_1\left(mol\right)\)
\(m_{ddNaOH10\%}=1,12V_2\left(g\right)\) \(\Rightarrow n_{NaOH10\%}=\dfrac{1,12V_2.10\%}{40}=2,8.10^{-3}V_2\left(mol\right)\)
\(\Rightarrow7,875.10^{-4}V_1+2,8.10^{-3}V_2=4,32\left(1\right)\)
Mà: V1 + V2 = 2000 (2)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}V_1\approx636\left(ml\right)\\V_2\approx1364\left(ml\right)\end{matrix}\right.\)