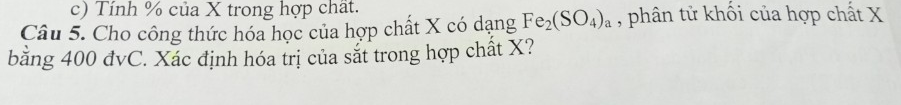Câu 17:
a) PTHH: \(Mg+2HCl\rightarrow MgCl_2+H_2\uparrow\)
b+c) Ta có: \(n_{Mg}=\dfrac{3,6}{24}=0,15\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{HCl}=0,3\left(mol\right)\\n_{H_2}=0,15\left(mol\right)=n_{MgCl_2}\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}V_{H_2}=0,15\cdot22,4=3,36\left(l\right)\\m_{ddHCl}=\dfrac{0,3\cdot36,5}{14,6\%}=75\left(g\right)\\m_{H_2}=0,15\cdot2=0,3\left(g\right)\\m_{MgCl_2}=0,15\cdot95=14,25\left(g\right)\end{matrix}\right.\)
d) Ta có: \(m_{dd}=m_{Mg}+m_{ddHCl}-m_{H_2}=78,3\left(g\right)\)
\(\Rightarrow C\%_{MgCl_2}=\dfrac{14,25}{78,3}\cdot100\%\approx18,2\%\)
a) $Mg + 2HCl \to MgCl_2 + H_2$
b) n H2 = n Mg = 3,6/24 = 0,15(mol)
V H2 = 0,15.22,4 = 3,36 lít
c) n HCl = 2n Mg = 0,3(mol)
=> m dd HCl = 0,3.36,5/14,6% = 75(gam)
d)
n MgCl2 = n Mg = 0,15(mol)
Sau phản ứng :
m dd = m Mg + mdd HCl - m H2 = 3,6 + 75 - 0,15.2 = 78,3(gam)
C% MgCl2 = 0,15.95/78,3 .100% = 18,2%
\(n_{Mg}=\dfrac{3.6}{24}=0.15\left(mol\right)\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
\(0.15.....0.3............0.15........0.15\)
\(V_{H_2}=0.15\cdot22.4=3.36\left(l\right)\)
\(m_{dd_{HCl}}=\dfrac{0.3\cdot36.5\cdot100}{14.6}=75\left(g\right)\)
\(m_{\text{dung dịch sau phản ứng}}=3.6+75-0.15\cdot2=78.3\left(g\right)\)
\(C\%_{MgCl_2}=\dfrac{0.15\cdot95}{78.3}\cdot100\%=18.2\%\)









 Mn ơi, giải hộ mik câu này với! Tối nay mik phải nộp rồi ạ! Cảm ơn mn nhiều lắm!
Mn ơi, giải hộ mik câu này với! Tối nay mik phải nộp rồi ạ! Cảm ơn mn nhiều lắm!