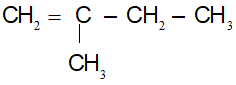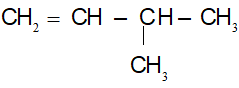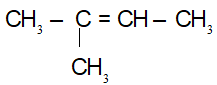\(n_{H_2O}=\dfrac{9}{18}=0,5\left(mol\right)\)
Bảo toàn H: nH(A) = 1 (mol)
=> mC = 7 - 1 = 6(g)
=> \(n_C=\dfrac{6}{12}=0,5\left(mol\right)\)
=> nC : nH = 0,5 : 1 = 1:2
=> CTPT: (CH2)n
Mà MA = 32.2,1875 = 70 (g/mol)
=> n = 5
=> CTPT: C5H10
CTCT:
(1) CH2 = CH – CH2 – CH2 – CH3
(2) CH3 – CH = CH – CH2 – CH3
(3)
(4)
(5)
\(n_{H_2O}=\dfrac{9}{18}=0,5\left(mol\right)\)
Bảo toàn H: nH(A) = 1 (mol)
=> mC = 7 - 1 = 6(g)
=> \(n_C=\dfrac{6}{12}=0,5\left(mol\right)\)
=> nC : nH = 0,5 : 1 = 1:2
=> CTPT: (CH2)n
Mà MA = 32.2,1875 = 70 (g/mol)
=> n = 5
=> CTPT: C5H10
CTCT:
(1) CH2 = CH – CH2 – CH2 – CH3
(2) CH3 – CH = CH – CH2 – CH3
(3)
(4)
(5)
(6)
(7)