Gọi $n_{Mg} = a(mol) ; n_{Fe\ pư} = b(mol) ; n_{Fe\ dư} = c(mol)$
$\Rightarrow 24a + 56b + 56c = 6,44(1)$
$Mg + 2AgNO_3 \to Mg(NO_3)_2 + 2Ag$
$Fe + 2AgNO_3 \to Fe(NO_3)_2 + 2Ag$
Theo PTHH : $n_{Ag} = 2a + 2b(mol)$
$\Rightarrow m_A = 108(2a + 2b) + 56c = 24,36(2)$
$Mg(NO_3)_2 + 2NaOH \to Mg(OH)_2 + 2NaNO_3$
$Fe(NO_3)_2 + 2NaOH \to Fe(OH)_2 + 2NaNO_3$
$Mg(OH)_2 \xrightarrow{t^o} MgO + H_2O$
$4Fe(OH)_2 + O_2 \xrightarrow{t^o} 2Fe_2O_3 + 4H_2O$
Theo PTHH : $m_{chất\ rắn} = 40a + 0,5b.160 = 7(3)$
Từ (1)(2)(3) suy ra : a = 0,035 ; b = 0,07 ; c = 0,03
$\%m_{Mg} = \dfrac{0,035.24}{6,44} .100\% = 13,04\%$
$\%m_{Fe} = 100\% - 13,04\% = 86,96\%$
b) $n_{AgNO_3} = n_{Ag} = 2a + 2b = 0,21(mol)$
$C_{M_{AgNO_3}} = \dfrac{0,21}{0,5} = 0,42M$






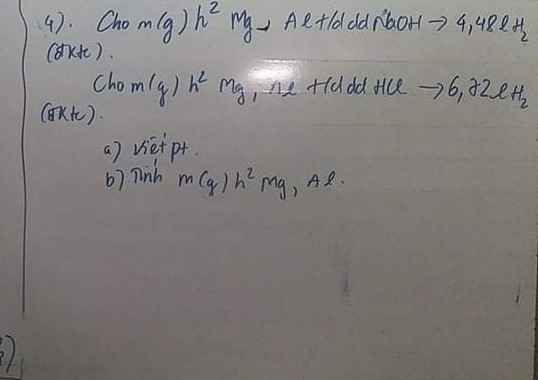









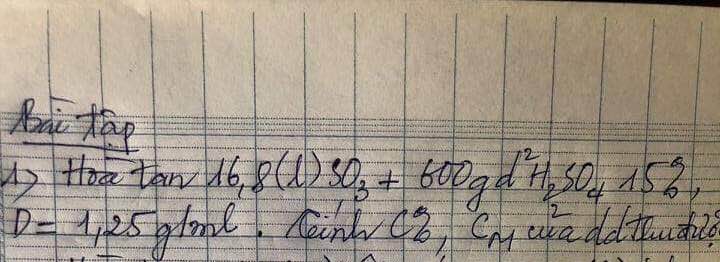





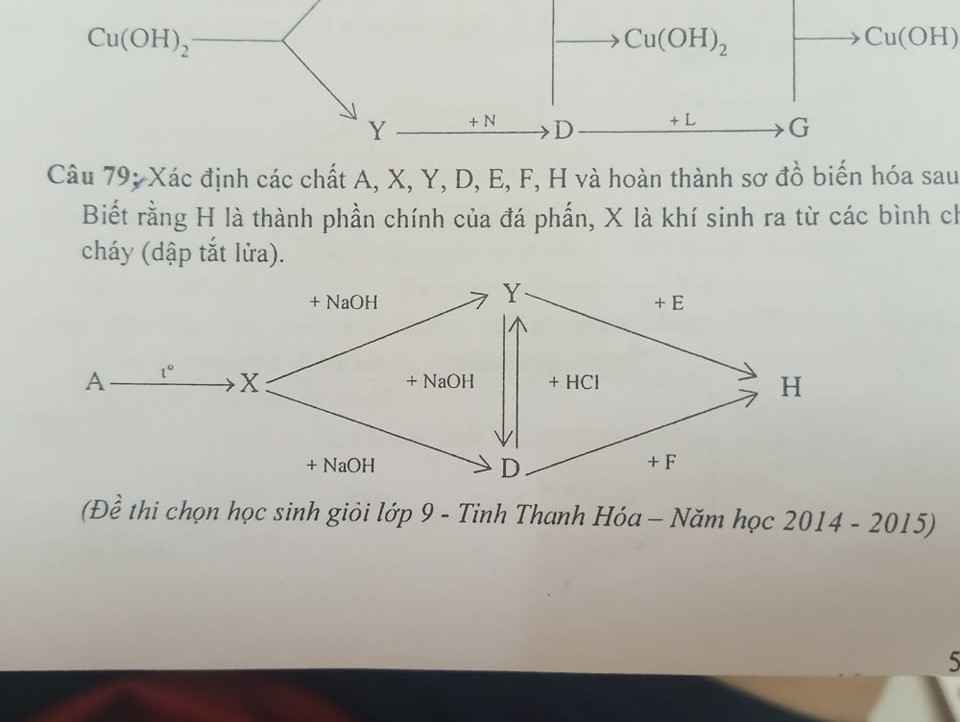 Cần gấp ạ
Cần gấp ạ