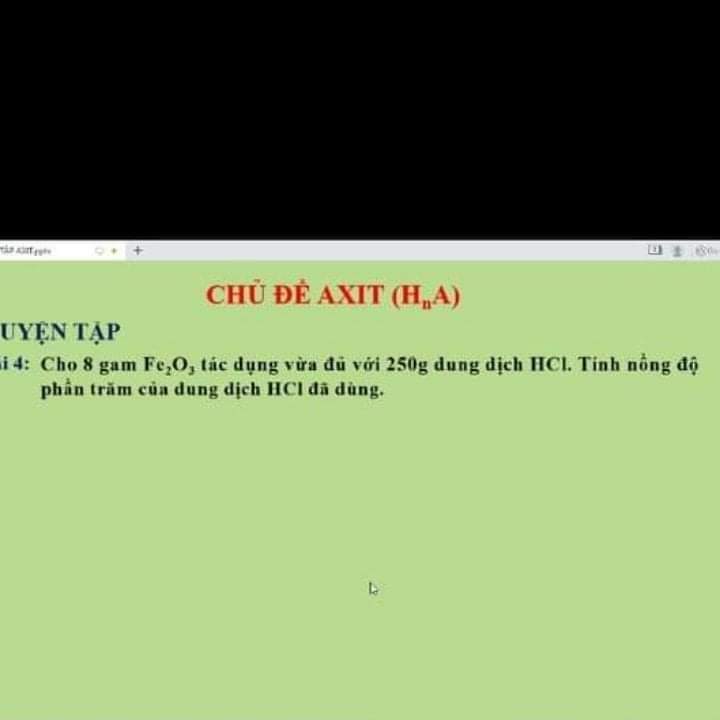
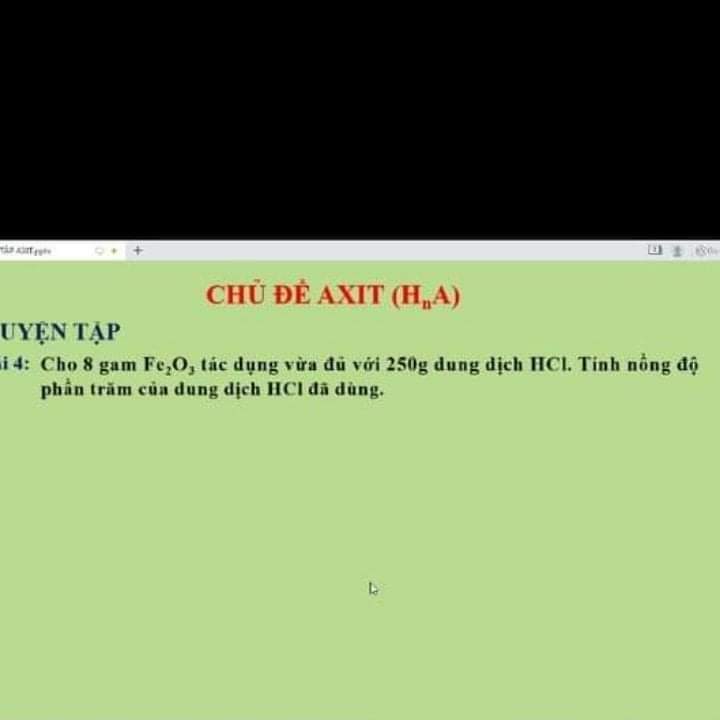
1)
a) Gọi \(n_{CuSO_4\left(pư\right)}=a\left(mol\right)\)
PTHH: Fe + CuSO4 ---> FeSO4 + Cu
a<---a------------->a-------->a
=> mtăng = mCu - mFe
=> 1,4 = 64a - 56a
=> a = 0,175 (mol)
=> \(\left\{{}\begin{matrix}m_{Fe}=0,175.56=9,8\left(g\right)\\m_{Cu}=0,175.64=11,2\left(g\right)\end{matrix}\right.\)
b) \(\left\{{}\begin{matrix}C_{M\left(CuSO_4.dư\right)}=\dfrac{0,2-0,175}{0,2}=0,125M\\C_{M\left(FeSO_4\right)}=\dfrac{0,175}{0,2}=0,875M\end{matrix}\right.\)
2)
\(m_{AgNO_3}=250.4\%=10\left(g\right)\)
\(m_{AgNO_3.giảm}=m_{AgNO_3\left(pư\right)}=10.17\%=1,7\left(g\right)\)
=> \(n_{AgNO_3\left(pư\right)}=\dfrac{1,7}{170}=0,01\left(mol\right)\)
PTHH: Cu + 2AgNO3 ---> Cu(NO3)2 + 2Ag
0,005<-0,01---------------------->0,01
=> mKL sau phản ứng = 10 - 0,005.64 + 0,01.108 = 10,81 (g)






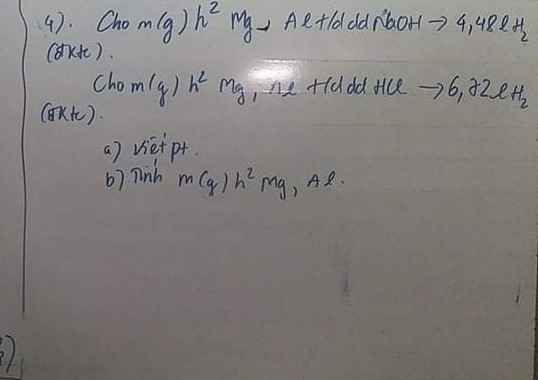






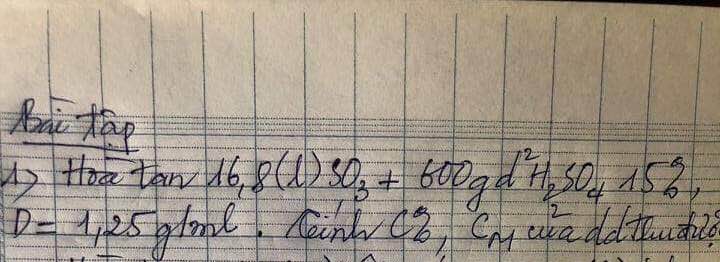





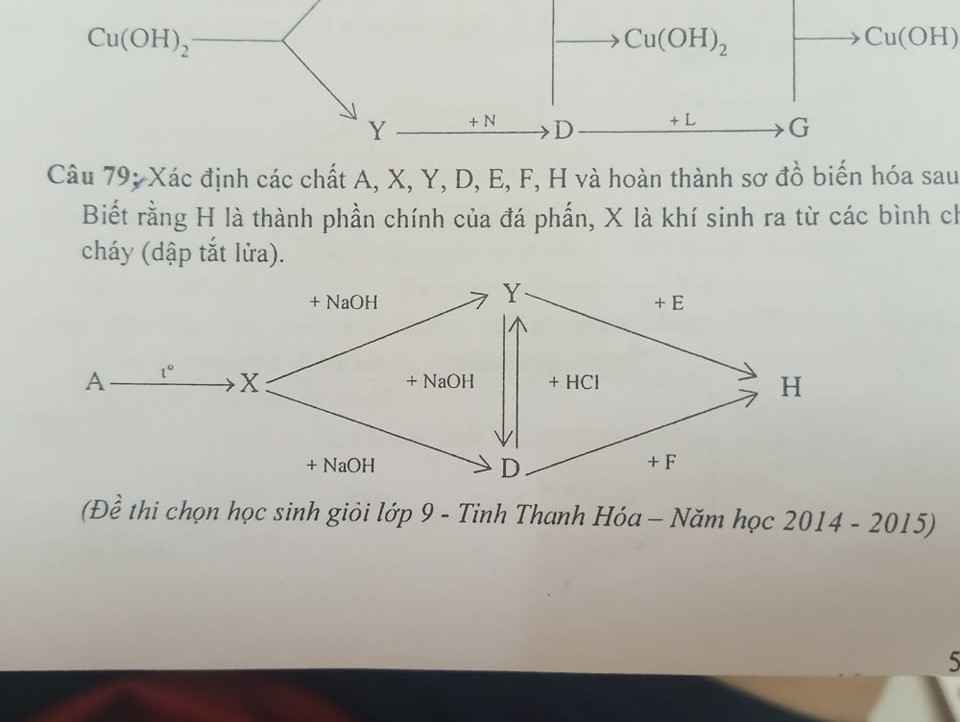 Cần gấp ạ
Cần gấp ạ