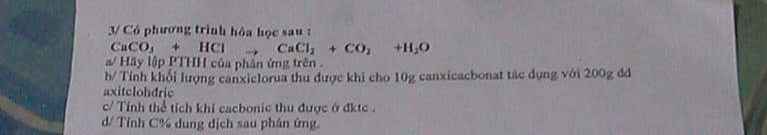Câu 3 :
\(CaCO_3+2HCl\rightarrow CaCl_2+CO_2+H_2O\)
\(n_{CaCO_3}=\dfrac{10}{100}=0.1\left(mol\right)\)
\(n_{CaCl_2}=n_{CO_2}=0.1\left(mol\right)\)
\(m_{CaCl_2}=0.1\cdot111=11.1\left(g\right)\)
\(V_{CO_2}=0.1\cdot22.4=2.24\left(l\right)\)
\(m_{\text{dung dịch sau phản ứng}}=10+200-0.1\cdot44=205.6\left(g\right)\)
\(C\%_{CaCl_2}=\dfrac{11.1}{205.6}\cdot100\%=5.39\%\)
a) CaCO3 + 2HCl → CaCl2 + CO2 + H2O
b) n CaCl2 = n CaCO3 = 10/100 = 0,1(mol)
=> m CaCl2 = 0,1.111 = 11,1(gam)
c) n CO2 = n CaCO3 = 0,1(mol)
V CO2 = 0,1.22,4 = 2,24(lít)
m dd sau pư = m CaCO3 + mdd HCl - m CO2 = 10 + 200 - 0,1.44 = 205,6 gam
C% CaCl2 = 11,1/205,6 .100% = 5,4%