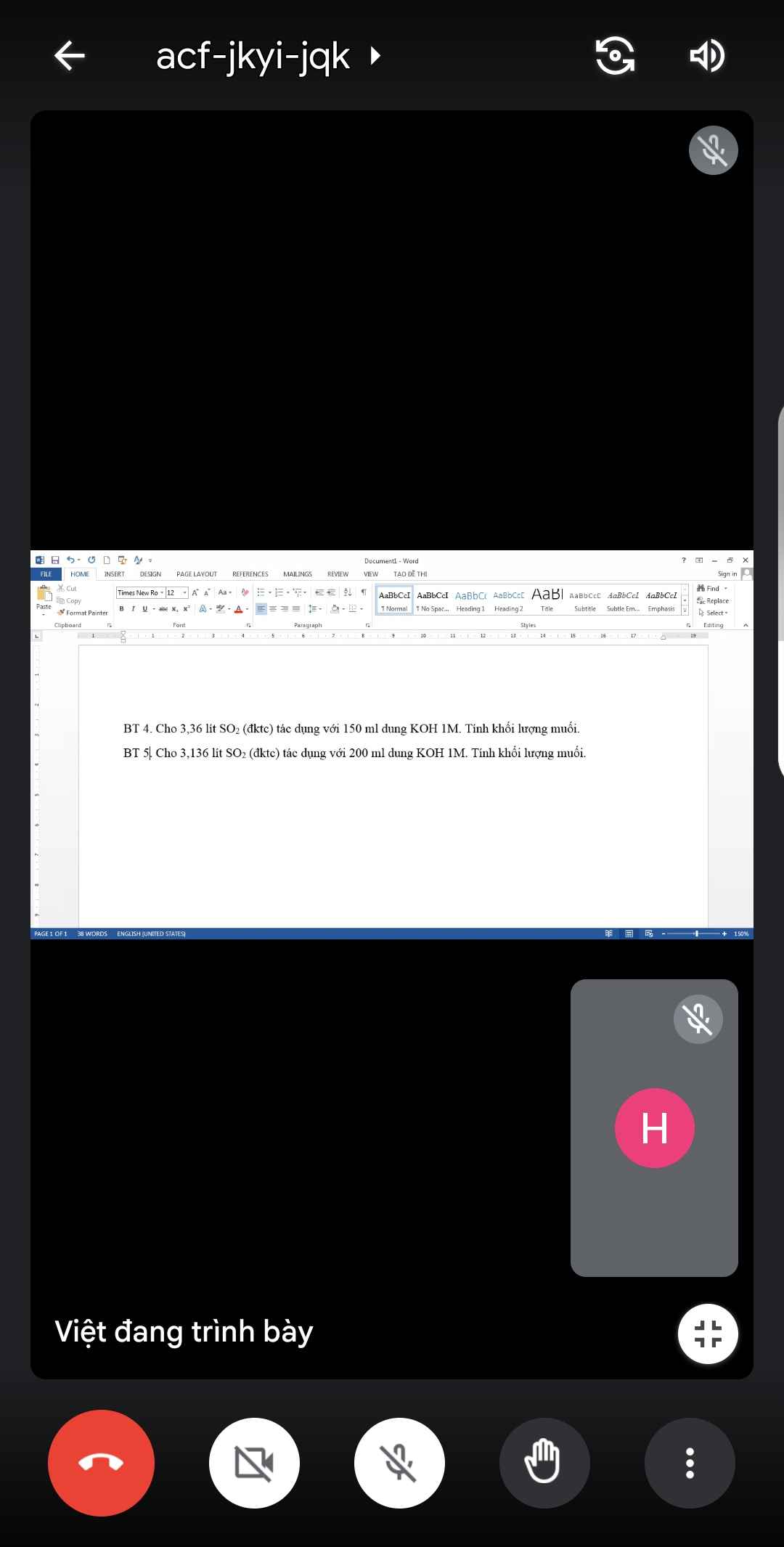
BT 4:
\(n_{SO_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
\(n_{KOH}=0,15.1=0,15\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{n_{KOH}}{n_{SO_2}}=\dfrac{0,15}{0,15}=1\) => Tạo muối KHSO3
PTHH: KOH + SO2 --> KHSO3
0,15------------->0,15
=> mKHSO3 = 0,15.120 = 18 (g)
BT 5:
\(n_{SO_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
\(n_{KOH}=0,2.1=0,2\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{n_{KOH}}{n_{SO_2}}=\dfrac{0,2}{0,15}=1,33\) => Tạo muối K2SO3, KHSO3
PTHH: 2KOH + SO2 --> K2SO3 + H2O
0,2-->0,1------->0,1
K2SO3 + SO2 + H2O --> 2KHSO3
0,05<---0,05-------------->0,1
=> Muối gồm \(\left\{{}\begin{matrix}K_2SO_3:0,05\left(mol\right)\\KHSO_3:0,1\left(mol\right)\end{matrix}\right.\)
=> mmuối = 0,05.158 + 0,1.120 = 19,9 (g)












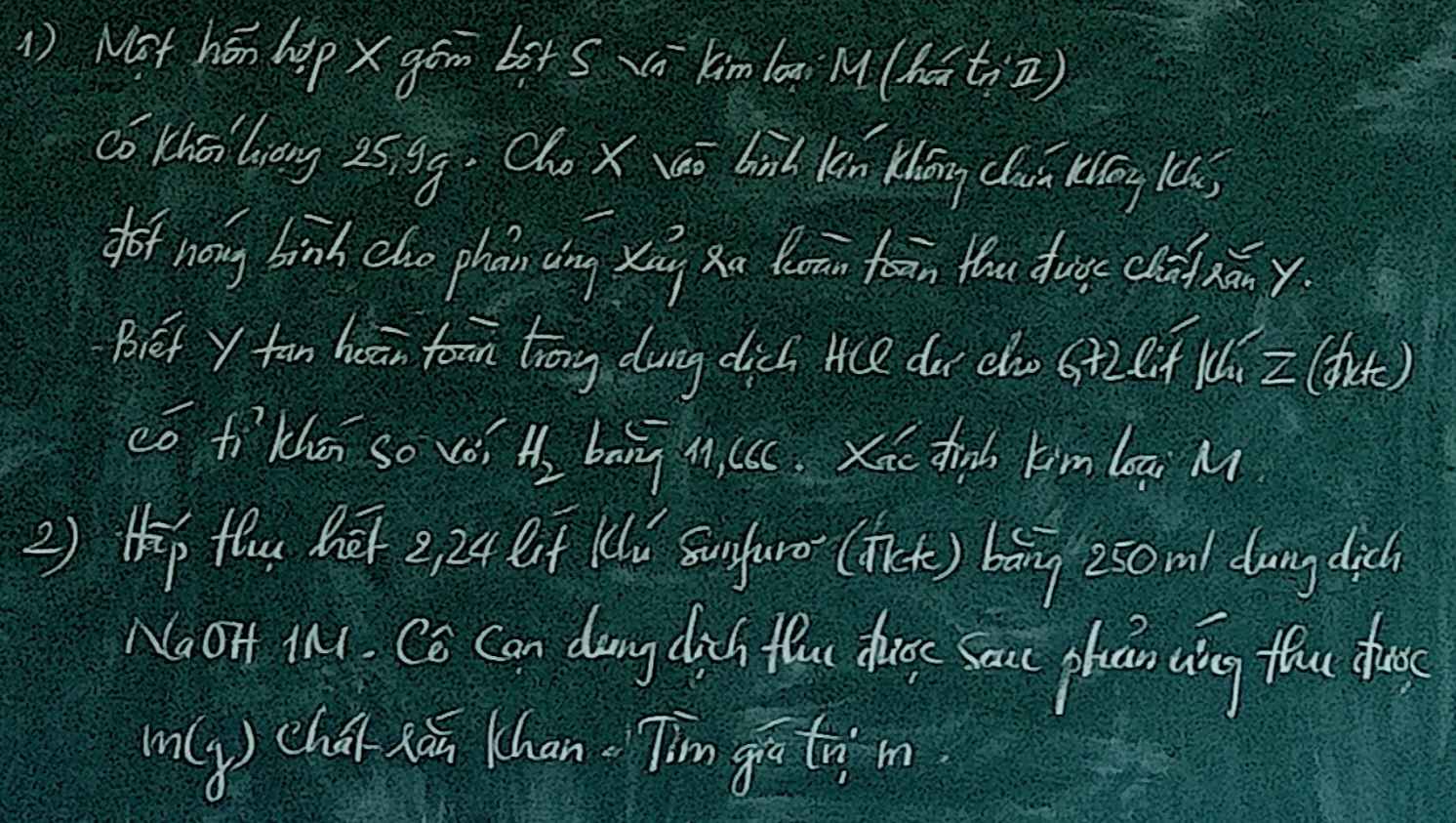

 Mn giúp mình câu 2 với ạ😭😭
Mn giúp mình câu 2 với ạ😭😭